Lowering homocysteine and adequate omega-3 status for slowing cognitive decline
VITACOG demonstrates adequate omega-3 status and lowering homocysteine with B-vitamins to slow brain atrophy and cognitive decline
Last year, dementia and Alzheimer’s disease surpassed cardiovascular disease as the leading cause of death in the UK. While CVD remains the leading cause of death globally and the largest contributor to global disease burden, neurodegenerative diseases are gaining market share rapidly.
Despite the significant financial incentives and resources poured into developing pharmacological strategies to combat neurodegenerative diseases, the results have been underwhelming, to say the least. The current medical management of neurodegenerative diseases is centered around slowing progression and treating symptoms.
As with most chronic diseases, prevention and early intervention seem rational under the confines of a justified risk-to-reward ratio, something I’ve discussed earlier here and here. However, unlike cardiovascular disease, our pharmacological strategies for neurodegenerative disease do not offer a lot in terms of prevention, mainly due to the complex etiology and lack of apparent causal factors.
An emerging perspective at the crossroad of neurology and cardiology is that “what’s good for the heart is good for the brain”. Indeed, high cardiorespiratory fitness, being strong, not smoking, being normotensive, having low lipids, not drinking too much alcohol, not being overweight, sleeping enough and well, and having good mental and emotional health are all predictive of better outcomes and less disease burden when it comes to both cardiovascular diseases and neurodegenerative diseases.
Despite the huge impact that efforts towards improvement in the beforementioned areas could yield, the growing burden of neurodegenerative disease might incentivize us to reach even further. For people who are at higher baseline risk, those with a family history of neurodegenerative disease or an unfavorable genotype, or a history of traumatic brain injuries, hedging one’s bets maximally under the confines of a justified risk-to-reward ratio does not sound ill-advised. The burden of evidence, however, is on the pro-interventionist, and hence we should have rigorous evidence to support claims beyond what is generally recommended and accepted as sound advice.
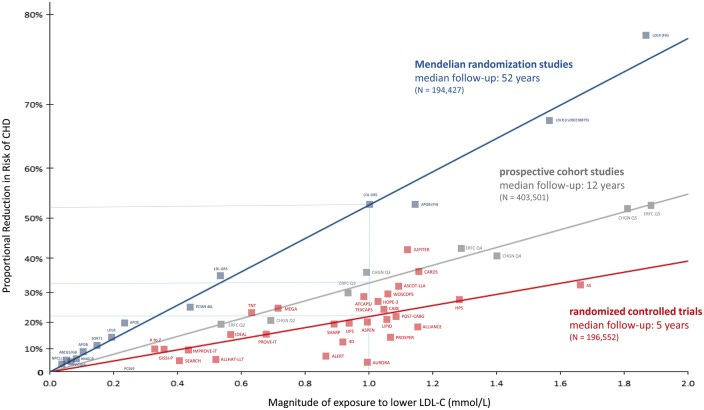
There are different levels of confidence that can be assigned to pharmacological interventions doing what they’re supposed to do. In contrast to treating an active disease, prevention can’t hang its hat on the process of “curing” a condition, which is the strongest evidence of the drug doing what it's supposed to do. For example, if a patient has pneumonia and gets treated with antibiotics, it’s clear that if they get cured of the pneumonia, the antibiotic was working. One could of course argue that they would have gotten cured without the antibiotic anyway, to which one in general can answer by showing the randomized controlled trials verifying the efficacy of the antibiotic. Another example is PDE5 inhibitors, meaning Viagra, Cialis, and so on. If a patient who has ED takes Viagra and regains the ability to get an erection, the drug is obviously working.
When it comes to taking a medication or supplement with the expectation of avoiding getting a disease, there’s no empirical evidence of “curing” to rely upon to support the practice. If a patient is prescribed a statin, for example, one could have high confidence that the statin does its job if there’s a way for us to quantify its effectiveness, i.e. a biomarker that gets impacted. Statins are a good example in that there is a way to measure their impact: measuring LDL-c, non-HDL-c, and/or apoB. Since apoB-100-bearing particles are known to be causal agents in the development of atherosclerosis, lowering them pharmacologically will likely result in less atherosclerosis, as is the case with using statins to reduce LDL-c, non-HDL-c, and/or apoB levels.1 The same holds for anti-hypertensives: high blood pressure is considered to cause cardiovascular disease and if a drug reduces blood pressure significantly, it very likely reduces major adverse cardiovascular events, i.e. our “biomarker” to follow-up on and target of treatment is blood pressure. Ultimately, the evidence in support of using statins or antihypertensives is rooted in randomized controlled trials demonstrating that they do not only lower the risk of major adverse cardiovascular events but do so safely and while reducing all-cause mortality.
Many biomarkers correlate positively with the development and progression of neurodegenerative diseases, but the causal nature of these is yet to be established. Already over a decade ago, however, David Smith and colleagues set out to determine if lowering homocysteine with B vitamins could impact brain atrophy seen in mild cognitive impairment and dementia, hopefully translating to slower cognitive decline with age.2 As Smith et al outline in VITACOG, elevated homocysteine levels are associated with accelerated brain atrophy and an increased risk of dementia, especially Alzheimer's disease, in many cross-sectional and prospective studies. I’ve written about homocysteine previously, so I will spare the biochemical details and move on to discussing VITACOG and its implications.
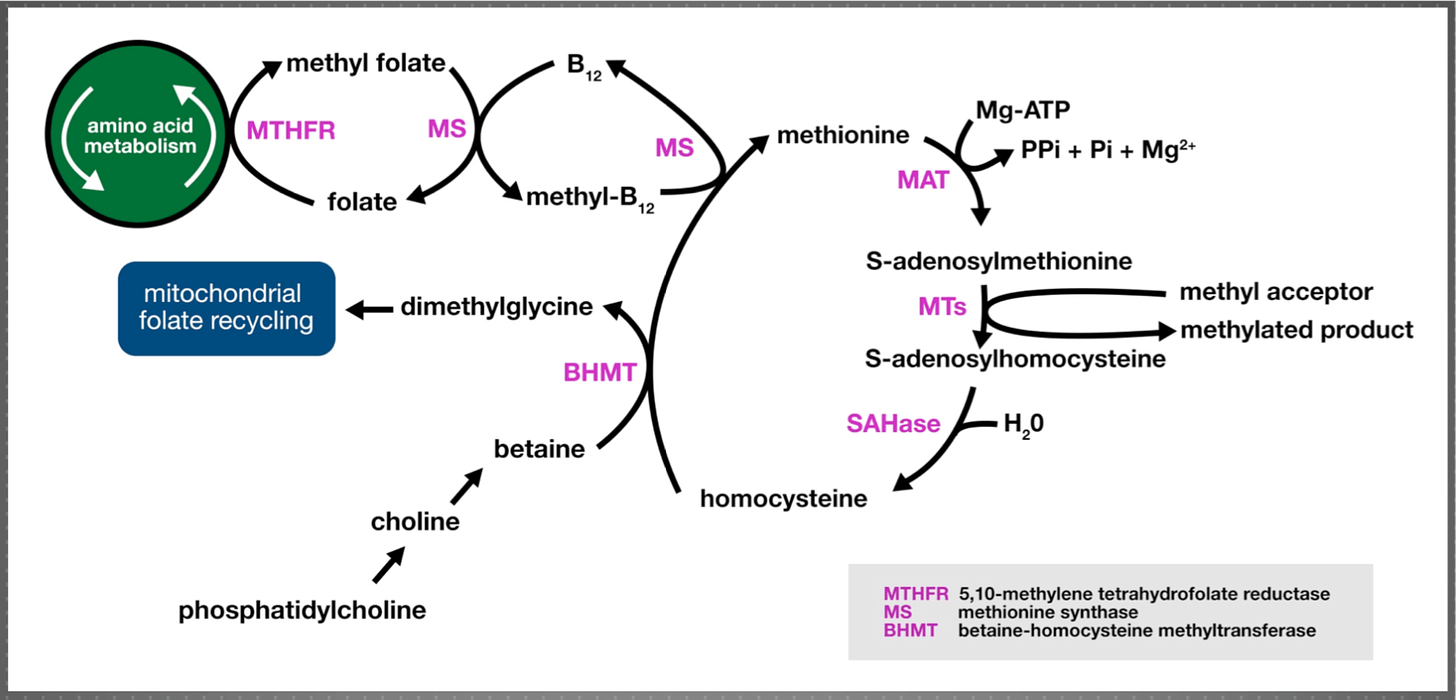
VITACOG was a single-center, randomized, double-blind, placebo-controlled trial investigating the effects that high-dose B vitamins can have on brain atrophy. B vitamins play a crucial role in the recycling of homocysteine and more specifically in one-carbon metabolism or the methylation cycle, and are thus essential for a wide variety of cellular processes. Elevated homocysteine is generally considered a sign of B vitamin deficiency, but even moderate elevations without signs of deficiency or inadequate B vitamin status are associated with both cardiovascular disease and neurodegenerative diseases. VITACOG sets out to establish the nature of this association and tries to clarify firstly if and how B vitamins can be used to prevent or slow the progression of cognitive decline, and secondly if homocysteine is generally a good measure of the effectiveness of the intervention.
271 individuals over 70 years of age, with mild cognitive impairment, were recruited to receive either high-dose folic acid, vitamins B6 and B12, or a placebo for 24 months. A subset volunteered to receive cranial MRI scans at the start and finish of the study to determine the rate of brain atrophy. The baseline homocysteine levels of both the placebo and treatment groups were 11.25 µmol/L. Folate levels and B12 levels rose by approximately 270% and 100%, respectively, in the treatment arm, and remained at baseline levels in the placebo arm.
Not surprisingly, homocysteine levels decreased by 22.5% in the treatment group and increased by 7.7% in the placebo group. Baseline homocysteine levels were associated with brain atrophy during the 24-month follow-up. As Smith et al concludes:
Treatment with B vitamins for 24 months significantly slowed the rate of brain atrophy. After adjustment for age, the rate of brain atrophy per year was 29.6% less in the active treatment group (0.76% [95% CI, 0.63–0.90]) compared to the placebo group (1.08% [0.94–1.22], P = 0.001)… If we confined the analysis to the biologically compliant subjects (n = 136), the effect of treatment was slightly greater with a reduction in atrophy rate of 31.1% in the active treatment group (rate of atrophy: 0.73% [0.57–0.88]) compared to the placebo group (1.06% [0.90–1.22], P = 0.004 after multi-adjusted analysis).
In the placebo group, homocysteine levels at baseline showed a striking positive relationship to the rate of atrophy (R2 = 0.24), whereas this association was absent in the active group.

These results imply that those with high baseline (t=0 months) homocysteine levels, who received and were compliant with the treatment, experienced the same amount of brain atrophy (in 24 months) as those with low baseline homocysteine levels receiving treatment. The amount of brain atrophy that the whole treatment group experienced was similar to those with low homocysteine in the placebo group, i.e. participants in the placebo group with high baseline homocysteine levels did experience more rapid brain atrophy than those with high baseline homocysteine and receiving B vitamins. The authors go on to describe the data in more detail than just “high” or “low” homocysteine:
In line with the interaction with baseline homocysteine described above, in participants with baseline homocysteine below the median, the active treatment was associated with 11.2% slower rate of atrophy, whereas those with baseline tHcy above median showed a 43.0% reduction in atrophy.
When tHcy was further divided into quartiles, there was no effect of treatment in those in the lowest quartile (tHcy ≤9.5 µmol/L), whereas there was a 53.3% reduction in rate of atrophy in those in the 4th quartile of tHcy (>13.0 µmol/L) treated with B vitamins vs. placebo.
Further, accelerated brain atrophy was observed in patients in the placebo group with a history of stroke or TIA (atrophy rate per year of 1.76% vs. 1.01%). This was not observed in the treatment group, where atrophy rates of 0.74% and 0.77% per year in those with a history of stroke vs. not were measured, meaning that the B vitamin treatment did “normalize” the accelerated brain atrophy associated with stroke and TIA. This has meaningful implications for post-stroke treatment and in patient with TIA, who hasn’t yet had a stroke.
Smith and colleagues go on to conclude:
…the treatment effect was greatest in those with the highest baseline level of homocysteine, with a reduction in atrophy rate of 53% in those in the top quartile of tHcy (>13.0 µmol/L). Notably, there was no effect of treatment on atrophy in those in the bottom quartile (≤9.5 µmol/L). The observation in the placebo group that the rate of atrophy was related to the baseline concentration of homocysteine is consistent with, and may explain, a number of cross-sectional studies reporting that regional brain atrophy is related to homocysteine. The results also fit with our earlier finding that raised homocysteine predicts the rate of shrinkage of the medial temporal lobe in patients with Alzheimer's disease.
It is tempting to suggest that this finding is consistent with the view that raised homocysteine is a direct cause of the atrophy. However, it does not exclude that homocysteine is only a marker for low-normal levels of the vitamins, which are themselves the causal factors.
Now, this study was not powered to detect how the treatment might impact the progression of cognitive decline, as it is not clear that brain atrophy translates to worse cognition linearly. Post hoc analyses, however, did observe the same factors to predict cognitive decline and brain atrophy and the authors conclude that in this specific population, “brain atrophy rate appears to be a major determinant of cognitive decline”.3
Taking a closer look at the post hoc analysis by Jager et al. reveals that the intention-to-treat group, overall, did only perform better on the CLOX test of executive function, but not on global cognition (MMSE), episodic memory (HVLT-R) or category fluency (CERAD) tests.
The odds of a correctly drawn item from CLOX1 at follow-up (24months), controlling for CLOX2 at follow-up and CLOX1 at baseline, as well as for age, education, APOE e4 status and sex, was 30% higher in B-vitamin-treated participants (P=0.015) relative to placebo.
However, when the participants in the intention-to-treat group are categorized based on baseline homocysteine, the results seem more promising:
The final model for HVLT-DR [episodic memory] revealed that participants taking placebo in the high tHcy group (≥ 11.3mmol/L) showed significant decline, whereas participants receiving B vitamins in the high tHcy group showed no significant decline.
The odds of correctly remembering a word from the list of 12 in the HVLT for a person in the high tHcy group at the end of the trial was 69% greater if they were taking B vitamins than if they were taking placebo.
B-vitamin treatment resulted in maintained per- formance in those with elevated tHcy, whereas placebo treatment was associated with decline in performance over time.
For the MMSE [global cognition], those in the high tHcy group who were treated with B vitamins were 1.58 times more likely to give a correct answer than those receiving placebo. In the low tHcy group, no significant difference was found in the odds ratio comparing treated and placebo.
For category fluency, in the high tHcy group, the average number of words at follow-up was 9.4% greater in those on treatment compared with those on placebo. In the low tHcy group, no significant difference was found when comparing treated with placebo.
So even though VITACOG was very likely underpowered to detect meaningful impacts of treatment on cognitive outcomes, a significant difference in performance was observed between treatment and placebo groups at follow-up, when considering the tertile with the highest homocysteine levels. This has clear implications for who to treat, and how aggressively.
The story doesn’t end here though. Oulhaj, together with Smith and colleagues, did further analysis of data from VITACOG to determine how the results of B vitamin treatment behaved in relation to omega-3 status.4 In this analysis, the outcomes of interest were episodic memory at the last follow-up and longitudinally, global cognition scores at the last follow-up, and clinical dementia rating at the last follow-up.
Subjects were ranked into tertiles based on omega-3 status. Tertiles showed “no difference in most demographic variables at baseline, including age, total years of education, smoking, blood pressure, BMI, and vitamin B12 concentrations. The highest tertiles of omega-3 were more likely to have a higher proportion of men, lower levels of tHcy, higher levels of folate, and lower levels of creatinine.”
As the authors go on to describe, the effect of the B vitamin treatment was significantly dependent upon the omega-3 status of the individuals:
The scores for HVLT-DR [episodic memory] at the last follow-up (24 months) increased in the B vitamin treated group across increasing tertiles of combined omega-3 fatty acids, whereas the scores in the placebo group showed negligible change.
When HVLT-DR was assessed in relation to tertiles of baseline plasma concentrations of DHA and of EPA separately, similar results were obtained and were particularly significant for DHA.

The TICS-M [global cognition] scores at the last follow-up (27 months) increased in the B vitamin treated group across increasing tertiles of combined omega-3 fatty acids, whereas the scores in the placebo group were not affected.
There was also a significant difference in the treatment effects between the 3rd and the 1st tertile of combined omega-3 fatty acids (difference = 2.85 points, p = 0.035) with higher scores for those in the 3rd tertile.
When TICS-M was assessed in relation to tertiles of baseline plasma concentrations of DHA and EPA separately, only higher concentrations of DHA significantly enhanced the cognitive effects of B vitamins. In the 3rd DHA tertile, there was a significant effect of B vitamin treatment on TICS-M and in the B vitamin treatment group there was a concentration-dependent effect of DHA with a 2.78 point higher TICS-M score in the 3rd tertile compared with the 1st tertile.

Clinical dementia rating follows in the same tract as the two previous measures:
In the 3rd tertile of baseline combined omega-3 fatty acids only 33% of those on B vitamin treatment had CDR scores >0 compared with 59% in the placebo group.
Also, in the 3rd tertile, B vitamins markedly reduced the risk of having CDR scores >0 (adjusted OR 0.31, 95% CI 0.10, 0.95; p = 0.043).
There was a trend for a concentration-dependent effect of combined omega-3 fatty acids: the proportion scoring >0 in the B vitamin group declined from 70% to 33% from the 1st tertile to the 3rd tertile (p = 0.053).
There was a concentration-dependent effect of DHA in the B vitamin group with a greater improvement in the 3rd than in the 1st tertile (p = 0.034). The effects of EPA on the CDR showed similar patterns but did not reach significance.
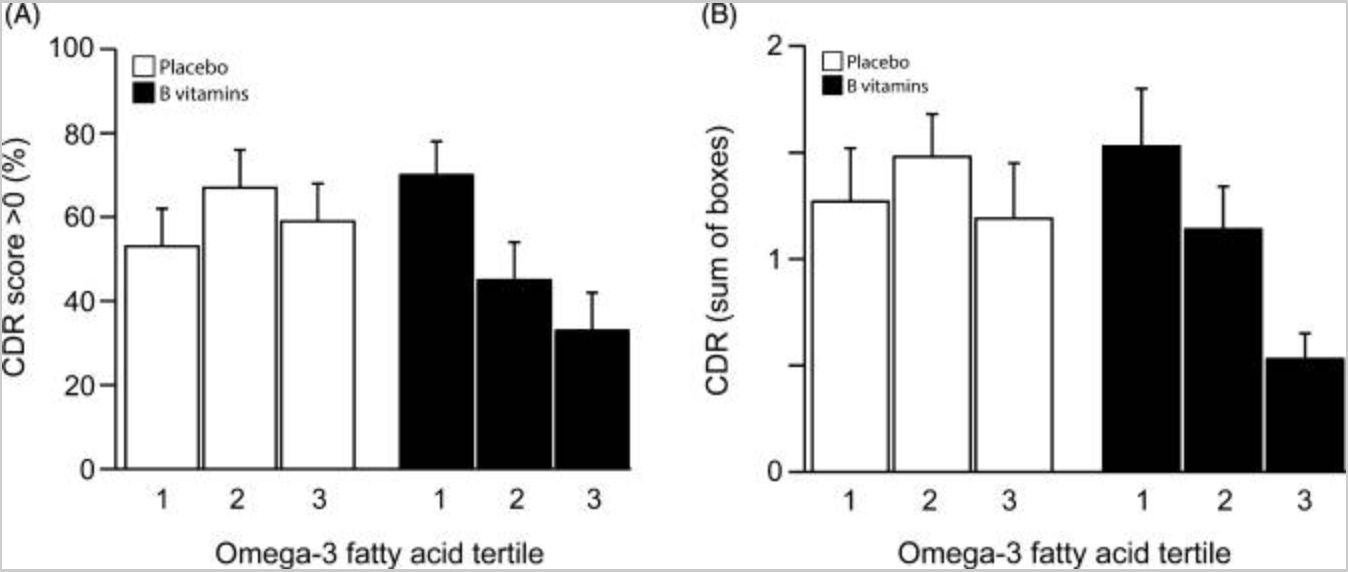
Oulhaj et al go on to conclude that “for participants with high baseline levels of omega-3 fatty acids, the B vitamin treated group performed better than placebo, while those with low baseline omega-3 fatty acids did not benefit from B vitamin supplementation. In general, the effects were more significant for DHA alone than for EPA or combined omega-3 fatty acids, especially for the HVLT-DR and TICS-M. However, due to the small number of participants and a considerable co-variation between DHA and EPA, the present data set is not ideal for separating the effects between the two fatty acids. Overall, DHA and EPA showed similar patterns in all cognitive outcomes. These results should be used for hypothesis generation and optimization of future trials.”
The results of VITACOG are certainly compelling and should raise our confidence that B vitamins, especially methylated B vitamins, and marine omega-3 fatty acids are effective in slowing cognitive decline. Furthermore, I believe the results of VITACOG validate using homocysteine measurements to predict the risk of cognitive decline, and also the effectiveness of B vitamin treatment. I’d also be confident that these compounds can be used “prophylactically”, although this might be stretching the evidence a bit.
In fact, it’s likely there will never be an adequately powered, randomized, placebo-controlled trial of sufficient duration looking at valid outcomes that investigate the effects of omega-3 FA:s and B vitamins for the purposes here. Hence, we need to adapt mentally to being “evidence-informed”, and be willing to extrapolate from the data and have confidence in our own judgment.
The treatments are of course not without their risk. It’s therefore of utmost importance to have a framework to rely upon when judging if an intervention is justified, and in the light of changing information, one needs to be willing to change their conclusions and actions to reflect the most current knowledge.


