Conflicting clinical outcomes on the pharmacological use of omega-3s for reducing the risk of adverse cardiovascular events
First in serie of trying to evaluate the risk to reward ratio of supplementing and pharmacologically using omega-3 fatty acids to reduce disease risk
There’s an ongoing debate in the circles of (preventative) cardiology about the benefits of pharmacological doses of omega-3 fatty acids (n-3s), especially eicosapentaenoic acid (EPA), as potential MACE-reducing agents. (Marine) omega-3 fatty acids, or “fish oils” in the form of EPA and DHA (docosahexaenoic acid) have been widely recommended by health practitioners and influencers as beneficial for cognition, joint, and muscle health, lowering inflammation, protective against cardiovascular disease, cancer, and aging, and so on. Despite the seemingly bullish attitude that many seem to maintain toward n-3s, the burden of proof remains on those claiming benefits, as the data is certainly not settled in terms of n-3s being net positive for specific objectives or even overall health.
In my previous entry Framework for pharmacological interventions and supplements, I discussed the general principles dictating if taking a drug or a supplement is justified. In general, we should consider 1) the objective we’re trying to achieve by taking an exogenous molecule, 2) the risk and possible downsides associated with taking, or not taking, said exogenous molecule, and 3) how to track the effects of taking said exogenous molecule. Analysis based on these three considerations is infinitely complicated and the only thing a practitioner can wish to gain is confidence in their hypothesis and practices. What level of confidence one finds satisfactory to make decisions and to intervene is ultimately dependent on the circumstances and the individual’s risk tolerance, and requires flexibility and willingness to change one’s attitude in light of new information.
N-3s have been written and talked about extensively by many practitioners and influencers in the health space. I will therefore refrain from talking about the biochemistry of n-3s and focus more on the “clinically” relevant data and studies, and steer the reader to the footnotes for a primer on the biochemistry.1 Due to the popularity of the topic, the public conversation around n-3s has been attracting people from different backgrounds with differing objectives. This ultimately results in conclusions made on varying amounts of confidence in the evidence and varying risk tolerances. Therefore, as stated earlier, it’s of utmost importance to articulate objectives clearly and assess the evidence in light of desired and undesired outcomes, and not hyperbolize one or the other. As is the case with all drugs and supplements, the discussion needs to get addressed in its full nuance, without bias, and as transparently as possible.
Marine omega-3 fatty acids in reducing major adverse cardiovascular events
I’ve previously discussed why assessing and reducing cardiovascular risk is one of the most important areas to focus on when it comes to extending both life- and healthspan. Epidemiological research has indicated n-3s to have beneficial effects on cardiovascular outcomes and mortality, which has spun up a whole industry of clinical research looking into the potential pharmacological properties and effects of n-3 fatty acids.2
One of the proposed mechanisms via which n-3s, and more specifically EPA, is thought to lower cardiovascular disease risk is via its effective lowering of triglycerides. In On triglycerides, I discussed some of the data pertaining to the pharmacological lowering of triglycerides and the associated challenges:
The distinction between primary causal factors (apoB) and secondary consequences of underlying [disease] processes is crucial when assessing the efficacy of an intervention. In high-risk patients, as stated earlier, apoB seems to be the desired primary target of intervention, while triglycerides in ranges below a significant risk of pancreatitis should be of secondary consideration.
In fact, due to the widely accepted view of apoB being the primary driver of atherosclerotic cardiovascular disease, EPA has mainly been studied for reducing residual risk, i.e. the risk that remains after effective control of LDL-c/apoB. This means that studies looking at EPA for reducing MACE are primarily in patients with acceptable levels of LDL-c/apoB or who are treated with statins or other lipid-lowering therapies. I point this out because it’s important not to major in the minors, and minor in the majors. Until we have comparative studies between apoB lowering agents and EPA it seems like the vast majority of benefits with regards to cardiovascular disease can be expected to come from reducing apoB, and what EPA has to offer makes a marginal difference only on the edges.
Nevertheless, even though it seems like the pharmacological lowering of triglycerides should be considered only secondary to lowering apoB, we cannot be fully confident that this holds for the pharmacological lowering of triglycerides with EPA. Fibrates, the primary class of drugs used to lower triglycerides, have failed to produce mortality benefits in similar cohorts as EPA has been studied in, and have therefore been questioned in terms of their utility in reducing marginal cardiovascular disease risk. In PROMINENT, pemafibrate reduce triglycerides by over 25% compared to baseline, but apoB and MACEs actually increased.3 As I described in On triglycerides, these results together with mendelian randomization studies have led many to conclude that lowering moderately elevated triglycerides is not as important as previously thought if other risk factors are managed. We should however not infer the failure of pemafibrate to translate to anything alike with regard to EPA. In fact, we have many reasons to think that the benefits from EPA are not only mediated via the lowering of triglycerides, but also by effects on inflammation, antioxidative, and endothelial and platelet function. Also here, until comparative studies between fibrates and EPA are conducted it’s not reasonable to assume that the risk-benefit profile is anything alike between these classes of compounds.
Clinical studies have tried to evaluate the potential benefits of EPA on residual cardiovascular disease risk. Japan EPA Lipid Intervention Study (JELIS), an open-label study enrolling 18,645 participants with hypercholesterolemia, randomized participants to receive either 1.8g of EPA + a fixed dose of a statin per day or only a fixed dose of a statin daily (control group).4 Both primary and secondary prevention participants were enrolled, and no pre-specified triglyceride levels were demanded. In fact, plasma triglyceride levels decreased (only) by 9% in the EPA group (vs. 4% in the placebo group) for participants with P-Trigly < 1.7 mmol/L at baseline, and significantly more in participants with elevated triglycerides at baseline. Total cholesterol and LDL-c saw marked reductions in both groups due to the statin intervention, with marginal increases in HDL-c.

The EPA + statin treatment was associated with a 19% relative risk reduction of adverse cardiovascular events compared to statin treatment alone:
The 5-year cumulative rate of major coronary events was 2.8% in the EPA group and 3.5% in controls, resulting in a significant relative risk reduction of 19% in the EPA group (p=0.011). EPA treatment was associated with a significant reduction of 24% in the frequency of unstable angina. The occurrence of coronary death or myocardial infarction was not significantly lower (22%) in the EPA group than in controls. The frequency of fatal or non-fatal myocardial infarction was not significantly reduced (23%) in the EPA group; however, that of non-fatal coronary events (including non-fatal myocardial infarction, unstable angina, and events of angioplasty, stenting, or coronary artery bypass grafting) was significantly lower (19%) in the EPA group than in controls.

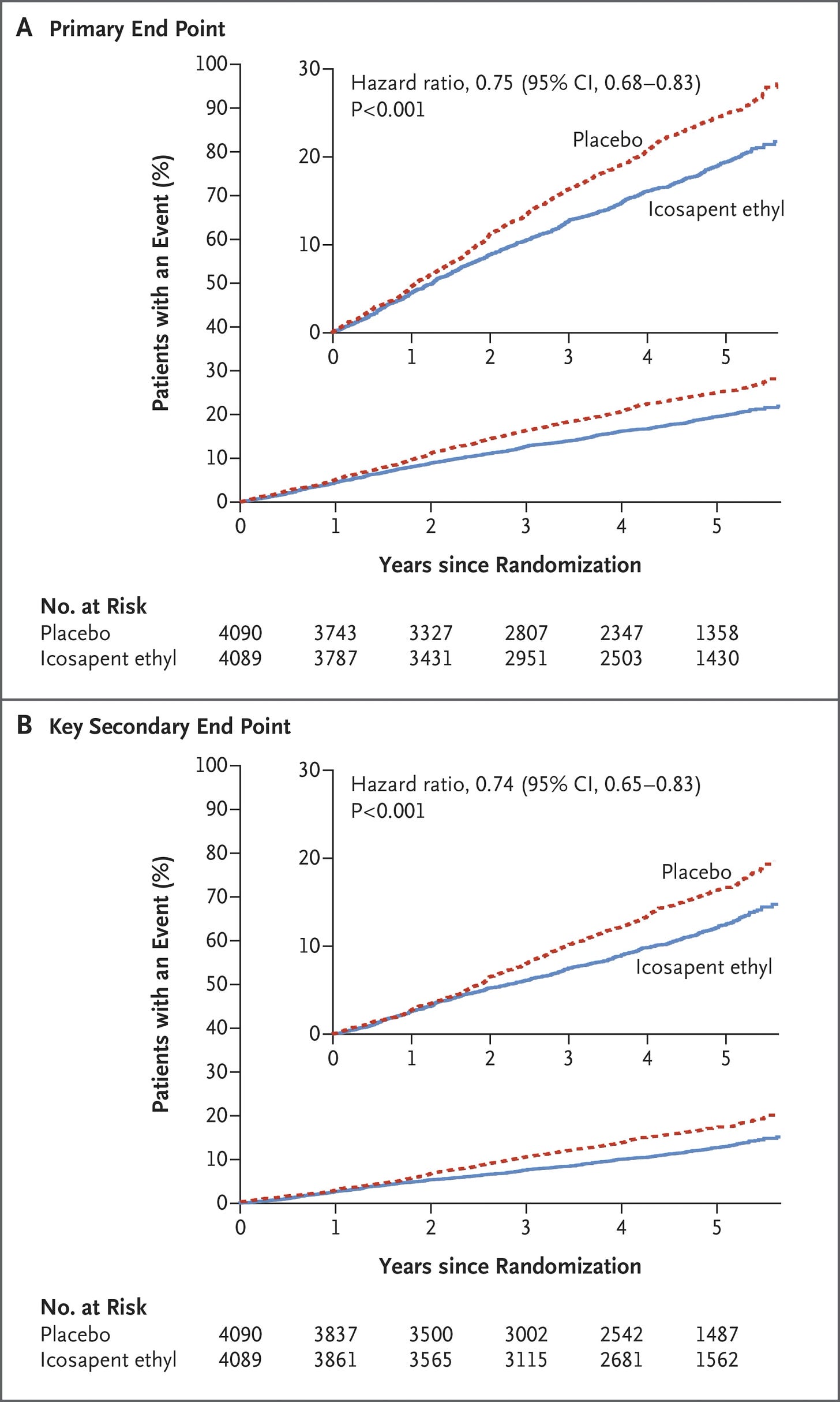
Reduction of Cardiovascular Events with Icosapent Ethyl–Intervention Trial (REDUCE-IT) is another clinical trial looking at the efficacy of EPA in reducing residual CVD risk.5 REDUCE-IT enrolled over 8,000 statin-treated participants with mildly to moderately elevated triglycerides, established CVD or diabetes, and at least one additional risk factor for CVD, and randomized them to receive 2g EPA 2x per day (4g/d tot.) or a mineral oil placebo. During the follow-up period of 4.9 years, triglycerides decreased by approximately 20% in the treatment group (from 216 mg/dL to 158 mg/dL). Interestingly, triglycerides increased by 2.2% at one-year follow-up in the placebo group but decreased by 3.7% and 1.5% at 4 and 5 years of follow-up, respectively. Non-HCL-c decreased by a little more than 4% from 118 mg/dL to 109 mg/dL in the treatment group and increased by 6.1% in the placebo group at 5 years of follow-up. ApoB also decreased by a marginal 2.5% in the treatment group and conversely increased by 7.8% and 4.5% at 2 years and "last visit" follow-up, respectively, in the placebo group. Also very interestingly, high-sensitivity CRP, which I've written about here, increased significantly in the placebo group. At both 2 years and "last visit" follow-ups a jump from 2.1 mg/L to 2.8 mg/L was noted in the placebo group, while a decrease from 2.2 mg/L to 1.8 mg/L was noted in the treatment group.

These previously mentioned changes to lipids and inflammatory markers have sparked some debate over the use of mineral oil as a placebo. Some argue that the mineral oil did not act as a placebo, but rather had pro-inflammatory features that led to the observed increases in hsCRP, apoB, and non-HDL-c.6 Whatever the case might be, REDUCE-IT did demonstrate a reduction in many relevant biomarkers, and did report reduced residual risk compared to statins alone (or in combination with mineral oil):
A total of 1606 adjudicated primary end-point events [5-point composite MACE: CV death, non-fatal MI, non-fatal stroke, hospitalization for unstable angina, and coronary revascularization] occurred. A primary end-point event occurred in 17.2% of the patients in the |EPA] group, as compared with 22.0% of the patients in the placebo group (hazard ratio, 0.75; 95% confidence interval [CI], 0.68 to 0.83; P<0.001), an absolute between-group difference of 4.8 percentage points (95% CI, 3.1 to 6.5); the number needed to treat to avoid one primary end-point event was 21 (95% CI, 15 to 33) over a median follow-up of 4.9 years.
A review on the subject of marine n-3s in cardiovascular disease offers further analysis of REDUCE-IT:7
Both post hoc and prespecified subgroup analyses revealed consistent event risk reduction with IPE [=EPA] regardless of CV disease history, including patients with prior MI, prior percutaneous coronary intervention, prior coronary artery bypass graft, or with varying kidney function based on estimated glomerular filtration rate…
…despite the baseline TG threshold required for enrollment in REDUCE-IT, IPE conferred a consistent risk reduction regardless of baseline or achieved TG levels, indicating that TG lowering itself does not explain the mechanism of event reduction in IPE-treated patients.
As the authors of the review point out, however, n-3s do not seem to be “risk-free”, which is something worth touching on later:
IPE [=EPA] treatment was associated with more serious adverse events in REDUCE-IT but similar to placebo. In particular, there were increased events related to hospitalization for atrial fibrillation (3.1% for EPA vs 2.1% for placebo, P = 0.004). However, this was not clinically significant as there were substantially lower rates of stroke with IPE (28% reduction in fatal or non-fatal stroke, P = 0.01). Bleeding rates overall were also low but slightly higher in subjects treated with IPE (2.7% vs 2.1%, p = 0.06), with no fatal bleeding events related to study drug. There was also no significant increase in gastrointestinal bleeding or hemorrhagic stroke events in adjudicated cases.
Before moving on to RESPECT-EPA, it’s worth noting that plasma EPA levels seem to be very important in these studies. In fact, the best predictor of outcomes in REDUCE-IT was EPA levels, which rose approximately 3.5x in the treatment group vs. the control group. What this means beyond the fact that the treatment group adhered to their protocol is not clear, but nevertheless, plasma EPA predicted events better than any traditional risk factor.
The benefits with IPE [=EPA] beyond TG lowering may indicate direct effects of EPA on progression of atherosclerosis not reproduced with low dose or mixed n3-FA preparations that include DHA in the formulation. Indeed, the best predictor for outcomes with IPE in REDUCE-IT was blood EPA concentrations as compared with other biomarkers of CV risk
RESPECT-EPA is a recent open-label randomized control trial investigating the effect of 1.8g of EPA per day paired with a statin vs. a statin alone in a Japanese population of participants with stable coronary artery disease and an EPA to AA (arachidonic acid) ratio under 0.4. The participants generally had more favorable biomarkers at baseline when compared to REDUCE-IT: higher plasma EPA and significantly lower hs-CRP levels. RESPECT-EPA saw no increase in lipids or hsCRP in the control group and achieved a small lowering of triglycerides and hs-CRP, and an increase in plasma EPA in the treatment group:
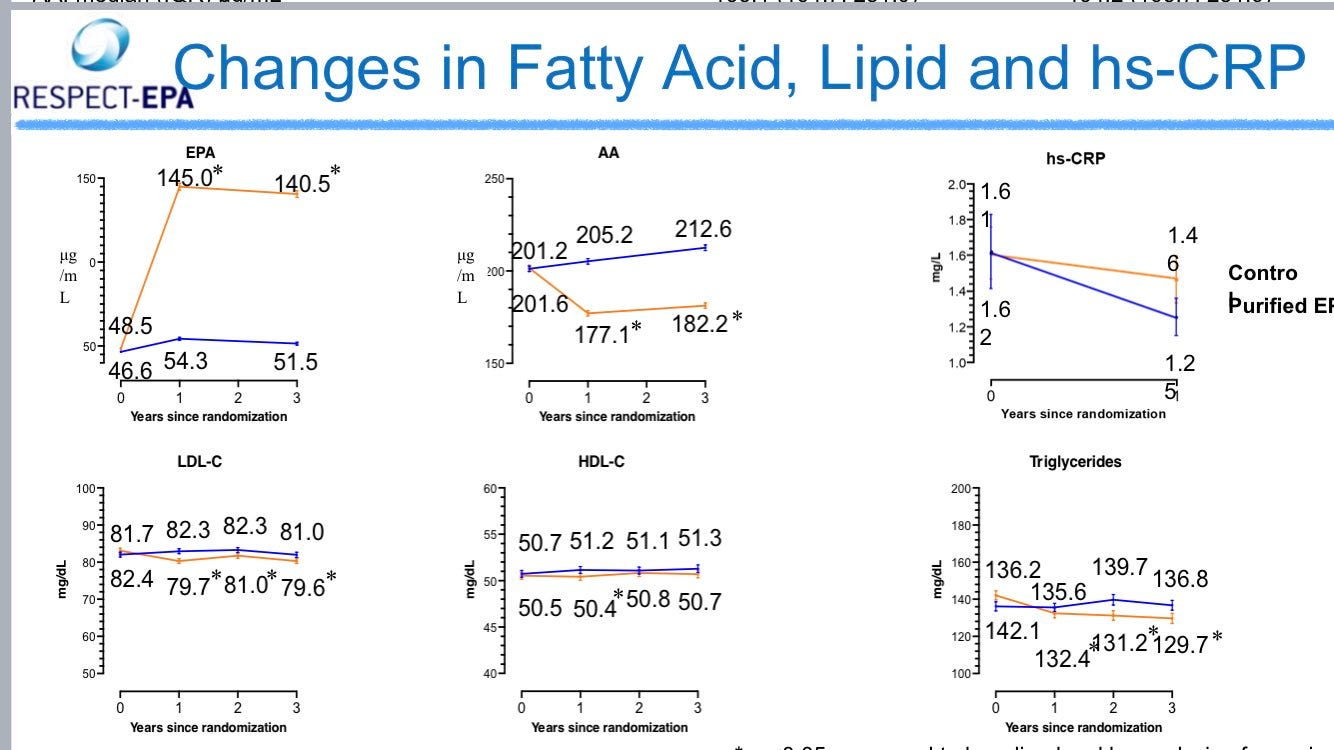
An article in published Practical Cardiology neatly summarizes the endpoints and results:
The study’s primary endpoints were a composite of cardiovascular death, nonfatal myocardial infarction, nonfatal cerebral infarction, unstable angina pectoris, and clinically indicated coronary revascularization. Secondary endpoints included composite events of CAD, composite events of stroke, and events related to death.
Within the RCT, the primary outcome occurred in 112 in the EPA group and 155 in the control group (hazard ratio [HR], 0.79; 95% confidence interval [CI], 0.62 - 1.00; stratified log-rank P-value = .055). Meanwhile, the secondary endpoint of coronary events occurred in 81 patients in the EPA group and 120 in the control group (HR, 0.73; 95% CI, 0.55 - 0.97; stratified log-rank P-value = .03).
The HR for all-cause mortality was 0.682 (95% CI, 0.385 - 1.208; stratified log-rank P-value = .1928) and the HR for cardiovascular mortality was 0.888 (95% CI, 0.625 - 1.262; stratified log-rank P-value = .5303).8
RESPECT-EPA caught the attention of many due to missing its primary end-points with a p-value of 0.055. The secondary end-point of composite events of CAD, composite events of stroke, and events related to death were hit with an HR, 0.73; 95% CI, 0.55 - 0.97; p-value = .03. There was no benefit with regard to cardiovascular and all-cause mortality.
Discussions on Twitter have led many to question the study design and the power to detect a benefit. Although the dose of EPA in RESPECT-EPA was smaller vs. REDUCE-IT, the plasma EPA levels achieved were similar. Some, therefore, seem to propose that the dose wasn’t the issue. Post-hoc analysis indicated that when the change in plasma EPA from baseline was greater than 30 μg/mL, there was a statistically significant decrease in primary end-point events, but these analyses have been put under scrutiny as statistical wizardry.


What’s more, is that EPA treatment was associated with significantly more adverse side effects, with increased rates of atrial fibrillation, bleeding, and gastrointestinal symptoms being the major ones. In RESPECT-EPA, participants in the treatment group experienced almost twice the rate of atrial fibrillation compared to controls (3.1% vs. 1.6%), but there didn’t seem to be any effect on bleeding. REDUCE-IT concluded similarly that the rate of atrial fibrillation increased in the treatment group (5.3% vs. 3.9%), as did the rate of peripheral edema (6.5% vs. 5.0%). Conversely, rates of anemia, diarrhea, and other gastrointestinal events were higher in the control group. Furthermore, in JELIS, gastrointestinal disorders, skin disorders, and abnormal liver function tests were higher in the EPA group.
|REDUCE-IT:] The rate of the prespecified adjudicated tertiary end point of hospitalization for atrial fibrillation or flutter was significantly higher in the icosapent ethyl group than in the placebo group (3.1% vs. 2.1%, P=0.004). The overall rates of serious adverse bleeding events that occurred while the patients were in the trial were 2.7% in the icosapent ethyl group and 2.1% in the placebo group (P=0.06), although there were no fatal bleeding events in either group; there were no significant differences between the icosapent ethyl group and the placebo group in the rates of adjudicated hemorrhagic stroke (0.3% vs. 0.2%, P=0.55), serious central nervous system bleeding (0.3% vs. 0.2%, P=0.42), or gastrointestinal bleeding (1.5% vs. 1.1%, P=0.15)
The results from JELIS, REDUCE-IT, and RESPECT-EPA lean towards EPA being beneficial for reducing residual risk. This is of course not without any caveats but is nevertheless giving us some signal of benefit. That being said, what one must note is that the doses and forms of EPA used in these trials aren’t anything like the ones recommended on commercial fish oil products. The before-mentioned clinical trials used doses in the range of 1.8-4 grams of EPA per day, which is generally two to four folds higher than the recommended dose on brand labels. Without getting into brand-specific issues, one can imagine it getting quite expensive, quite fast, if one were to consume 4x the daily recommended dose, which might be what is required for desired outcomes.
Furthermore, commercial products are generally sold as EPA + DHA combinations. The STRENGTH Randomized Clinical Trial evaluated the effects of an EPA + DHA formulation on residual CVD risk in a similar fashion as REDUCE-IT.9 13,078 participants with elevated triglycerides and known cardiovascular disease risk factors, including diabetes, were randomized to receive a statin and 4g of EPA + DHA daily or a statin in combination with a seemingly inert corn oil preparation.10 Despite a significant reduction of 19% in triglycerides in the n-3 treatment group, the trial was prematurely halted "based on an interim analysis that indicated a low probability of clinical benefit of omega-3 CA vs the corn oil comparator." Also, adverse events were significantly higher in the treatment group (22.2% vs 12.9%), with almost one in four participants experiencing gastrointestinal adverse events (24.7% vs. 14.7%).
STRENGTH is in many ways perplexing and I'm personally a bit disappointed that the study was halted early. The changes to biomarkers observed in the treatment group were in general favorable, with triglycerides decreasing by 19% in the treatment group vs. the control (−19.0% vs −0.9%), hs-CRP decreasing −20.0% vs −6.3%, non-HDL decreasing −6.1% vs −1.1%, and HDL-c increasing 5.0% vs 3.2%. This leads me to think that halting the study might have been premature, or that the study period would have been too short to detect potential benefits. That being said, we should be careful when making such assumptions. While it generally is desirable to see biomarkers move in the direction they did in the treatment group, it's not clear that changes translate over to event or mortality benefits.
The ASCEND Study Collaborative Group has also investigated the benefits of n-3 fatty acids for the primary prevention of cardiovascular events in diabetic participants.11 15,480 participants were randomized to receive 1g of EPA+DHA daily with no requirement of adherence to statin treatment and followed for 7.4 years. With regard to the primary and secondary outcomes, ASCEND failed to demonstrate benefits:
Serious vascular events occurred in 689 patients (8.9%) in the fatty acid group and in 712 patients (9.2%) in the placebo group (rate ratio, 0.97; 95% CI, 0.87 to 1.08; P=0.55), with no evidence of effects emerging with longer follow-up. There was also no significant between-group difference in the secondary outcome of serious vascular events or revascularization, which occurred in 882 patients (11.4%) in the fatty acid group and in 887 (11.5%) in the placebo group (rate ratio, 1.00; 95% CI, 0.91 to 1.09).
The most plausible explanation for this null finding is the lackluster dose of n-3s used. The trials with EPA-only formulations have clearly shown that quite large doses are needed to establish benefits, which is also now supported by lower doses being ineffective.
The proposed benefits of n-3s are not only mediated by favorable changes in lipids but also via modulation of inflammatory pathways. A previously cited review by Sherratt et al. on the topic of n-3s role in cardiovascular disease provides some insight into how n-3s impact inflammatory pathways: cells incorporate n-3s into their membranes, from which they can be released and used as substrates to generate specialized pro-resolving mediators by the enzymes COX and LOX, i.e. molecules that work to resolve inflammation. COXs and LOXs are also producing anti- and pro-inflammatory factors, as well as platelet activators, thromboxanes, and vasoconstrictors from omega-6 fatty acids, such as arachidonic acid. By competing with COX, n-3s reduce the generation of these pro-thrombotic and -inflammatory factors and promote the generation of factors that work to resolve inflammation and restore homeostasis after inflammatory processes. This should theoretically reduce chronic inflammation and contribute to endothelial health. Furthermore, EPA and DHA also promote endothelial-dependent NO release, contributing to increased vasodilation and vascular compliance.
Let’s now remind ourselves of the framework for pharmacological and supplemental interventions. Firstly, what are we trying to achieve by intervening with a supplement or pharmacological agent? Secondly, what does the risk-to-reward calculation look like for the specific exogenous agent? Lastly, how can one monitor the effectiveness of the intervention?
Cardiovascular disease is the leading cause of death worldwide, and also a big contributor to hospitalizations and decreased quality of life. Therefore, every strategy that tries to improve lifespan and healthspan has to contend with cardiovascular disease risk and management. Trials on n-3s have indicated potential benefits with regard to cardiovascular risk, and since n-3s, in general, are highly accessible as over-the-counter supplements, they are worth investigating as risk-reducing agents. It’s actually the fact that n-3s are so accessible, and also seem to be somewhat efficacious, which makes them one of the more popular supplements out there.
The risk-to-reward (r/r) calculation with n-3s is not straightforward. The most obvious “risk” is the financial cost of n-3s. A high-quality, over-the-counter n-3 preparation in adequate doses (based on the clinical trials) will cost about $1.50 to $3.00 per day. That’s quite expensive for a supplement, with $3 per day being $90 per month and over $1000 per year. Secondly, the increased risk of atrial fibrillation observed in the trials is to be taken seriously. Even though the absolute rates in REDUCE-IT and RESPECT-EPA were low (3.1% in both), the elevation is 1.5-2x compared to controls. Atrial fibrillation increases mortality by approximately 2x.12 The adverse gastrointestinal events can range from mild to severe, but this is less of an issue since the subject can easily discontinue in case of overwhelming symptoms. That’s less true for atrial fibrillation, which can be relatively asymptomatic and lead to fatal consequences. Moreover, patients who are at high risk for atherosclerotic cardiovascular disease are also more prone to atrial fibrillation, so the r/r is not trivial. Beyond the financial cost and side effects it’s worth considering interactions with other pharmacological agents and supplements. Due to the effect n-3s have on clotting, for example, concordant use with anticoagulants might be ill-advised.
Now, the benefits of n-3s on cardiovascular health aren’t that obvious either. JELIS, REDUCE-IT, and RESPECT-EPA did find EPA to lower residual risk, but as described earlier, not without caveats. STRENGHT and ASCEND did not find EPA + DHA preparation to be beneficial, despite seemingly positive changes to biomarkers. It’s important to remember that the observed benefits of EPA were mostly in combination with statins. This should raise suspicions about EPA’s effectiveness alone vs. in combination with statins, and we should be careful about overinterpreting results to be solely due to EPA treatment.
That being said, and on the topic of biomarkers, what stands out from the before mentioned studies is the predictive value of plasma EPA levels, as briefly mentioned earlier. In REDUCE-IT “the median percentage change in EPA levels from baseline was 393.5% at Year 1 (Year 1−5 range: 393.5–478.6%) which was sustained to Year 5 (p<0.0001). Over 5 years, the median change in serum EPA levels from baseline in the placebo arm ranged from -12.8% to 2.8%.”13
…similar [to triglycerides, approx. 2% of the observed 25% risk reduction] limited impact was found on other biomarkers including LDL cholesterol, HDL cholesterol, non-HDL cholesterol, apolipoprotein B, high-sensitivity C-reactive protein, and remnant-like particle cholesterol. Covariate analysis of on-treatment serum EPA levels found that they accounted for almost all of the 25% relative risk reduction in the primary endpoint, with an adjusted covariate HR of 1.03 (95% CI: 0.91−1.16). Therefore, serum EPA levels are highly correlated to atherosclerotic CVD risk reduction.
High EPA levels were associated with significant decreases in the HR of the primary endpoint, the key secondary endpoint, CV death, and total mortality (p<0.001). These data suggest that the higher the serum EPA level, the greater the CV benefit observed. Similar correlations were also noted in fatal and nonfatal MI, fatal and nonfatal stroke, coronary revascularisation, and unstable angina requiring hospitalisation (p<0.001)
This certainly raises my interest in looking at plasma EPA levels, at least when considering residual risk. I’ve spoken about other relevant biomarkers earlier in length, so suffices to say that the changes in apoB/non-HDL-c, HDL-c, triglycerides, and hs-CRP all seem to move in favorable directions. Despite this, whether accomplishing these changes by taking n-3s is beneficial or not for outcomes remains undecided.
In this post, I didn’t get into the nutritional epidemiology supporting fish intake. I’m quite confident that eating fatty fish regularly in the context of an otherwise nutrient-dense and minimally processed-food diet is health-promoting, or at least not harmful (beware of heavy metals though). The literature seems to indicate that plasma EPA levels do yield predictive value when it comes to cardiovascular disease, but whether attaining sufficient EPA levels via diet alone vs. from supplements is possible or net-beneficial remains unanswered. In light of the current literature and with cardiovascular prevention as the main objective, I’d say that increasing fish intake to attain and maintain “good” or “high” plasma EPA levels seems advisable. Whether one can tolerate the other risks associated with at least supplemental use of n-3s comes down to the individual, and should definitely be of secondary consideration when compared to lipid-lowering therapies or glucose/insulin regulation, for example. Pharmacologically or supplementally raising plasma EPA levels seems to have some marginal benefit in at least high-risk populations, and might therefore have its place in a carefully constructed regime that puts emphasis first and foremost on lifestyle changes and pharmaceutical intervention, and only thereafter, if risks are considered to be elevated, should n-3 therapies be considered.
I’d remind the reader of the fact that n-3s have effects that far exceed those on the cardiovascular system. The benefits with regard to cognition and other domains are not to be ignored and could potentially change the r/r drastically. In the future, I hope to have the time to go through the literature in a more all-encompassing fashion, with more emphasis on domains beyond the cardiovascular system.
Lastly, I’m not a physician (yet), and nothing in this post should be interpreted as medical advice and is for general information purposes only. You should not delay seeking medical advice for any problem that you might have.
I hope you found this post interesting and thought-provoking. Please like and share if you did, it’s the best way to support my work.
Take care,
Daniel
mean age, 62.5 years; 35% women; 70% with diabetes; median low-density lipoprotein [LDL] cholesterol level, 75.0 mg/dL; median triglycerides level, 240 mg/dL; median HDL-C level, 36 mg/dL; and median high-sensitivity C-reactive protein level, 2.1 mg/L
https://www.kaypahoito.fi/hoi50036#s7




