A recent study, PROMINENT: Triglyceride Lowering with Pemafibrate to Reduce Cardiovascular Risk, published in the NEJM, investigated the effects of Pemafibrate on cardiovascular events in patients with type 2 diabetes and mixed dyslipidemia. Pemafibrate belongs to a class of drugs called fibrates, whose main effect is to lower plasma triglycerides by acting as peroxisome proliferator-activated receptor (PPAR) modulators. PPARs are a complex topic on their own, and I’m not sure the scientific community fully understands PPARs yet, but suffices to say that they work as nuclear receptors, modulate the transcription of numerous genes, have as their ligands fatty acids, eicosanoids, many vitamins, and numerous drugs, and play important roles in fatty acid and glucose metabolism. Pemafibrate is a selective peroxisome proliferator-activated receptor α modulator used to lower plasma triglycerides and raise HDL-c.
PROMINENT investigated the impact of pemafibrate on cardiovascular events in a population of mostly obese, diabetic, hypertensive, and dyslipidemic patients with ongoing antihypertensive and statin treatment. 67% of the cohort had suffered from at least one previous adverse cardiac event.

The effects of pemafibrate on lipids were significant:
As compared with placebo, the effects of pemafibrate on lipid levels at 4 months were −26.2% for triglycerides, −25.8% for very-low-density lipoprotein (VLDL) cholesterol, −25.6% for remnant cholesterol (cholesterol transported in triglyceride-rich lipoproteins after lipolysis and lipoprotein remodeling), −27.6% for apolipoprotein C-III, and 4.8% for apolipoprotein B.
But despite these changes, the trial failed to reduce events vs. placebo:
After randomization, a primary end-point event occurred in 572 patients in the pemafibrate group and 560 patients in the placebo group (hazard ratio, 1.03; 95% confidence interval [CI], 0.91 to 1.15; P=0.67). With respect to the key secondary end point of myocardial infarction, ischemic stroke, unstable angina warranting urgent coronary revascularization, or death from cardiovascular causes, the corresponding numbers were 432 patients in the pemafibrate group and 417 patients in the placebo group (hazard ratio, 1.04; 95% CI, 0.91 to 1.19).1
Although, as compared with placebo, pemafibrate lowered triglyceride levels by 26.2% and increased HDL cholesterol levels by 5.1%, it did not reduce the incidence of events that made up the primary efficacy end point (a composite of nonfatal myocardial infarction, ischemic stroke, coronary revascularization, or death from cardiovascular causes). Patients who received pemafibrate had a higher incidence of renal events and venous thromboembolism and a lower incidence of nonalcoholic fatty liver disease than those who received placebo.2
The results of this study have been the source of some discussion in the community. The triglyceride to HDL ratio (TG/HDL) has traditionally been considered, by some, as an important predictor of CVD, mainly because high triglycerides and low HDL make up two of the five defining features of metabolic syndrome, and maybe more importantly, give us insight into early manifestations of insulin resistance.3 The results from PROMINENT and clinical trials done on HDL-increasing therapies seem to indicate firstly that pharmacological modulation of TG/HDL does not result in decreased CVD. Secondly, plasma triglycerides do not seem to be directly causal in the development of CVD. Thus, in contrast to previous notions, the burden of proof shifts to those who speak of triglycerides as a primary meaningful metric when assessing the risk of CVD.
The question then becomes: are triglycerides independently predictive of cardiovascular disease? If so, at what level and to what degree? And if not, what are the factors that really drive cardiovascular disease, and how do triglycerides relate to these?
Triglycerides gained their ground as a risk factor for atherosclerosis mainly based on clinical observations, epidemiological studies, and trials with triglyceride-lowering therapies. In a review named Triglycerides and cardiovascular disease4 Nordestgaard et al. describe how meta-analyses of epidemiological studies found increased triglycerides to be highly correlated with CVD:
…three studies based on the Copenhagen City Heart Study and the Women's Health Study suggested that increasingly raised non-fasting triglycerides were strongly associated with increasing risks of myocardial infarction, ischaemic (coronary) heart disease, ischaemic stroke, and all-cause mortality. In women, and for concentrations higher than 5 mmol/L versus those less than 1 mmol/L, the age-adjusted risk was increased 17 times for myocardial infarction, 6 times for ischaemic heart disease, 5 times for ischaemic stroke, and 4 times for all-cause mortality during 27–30 years of follow-up. For men, the corresponding risk increases were 5 times, 3 times, 3 times, and 2 times.
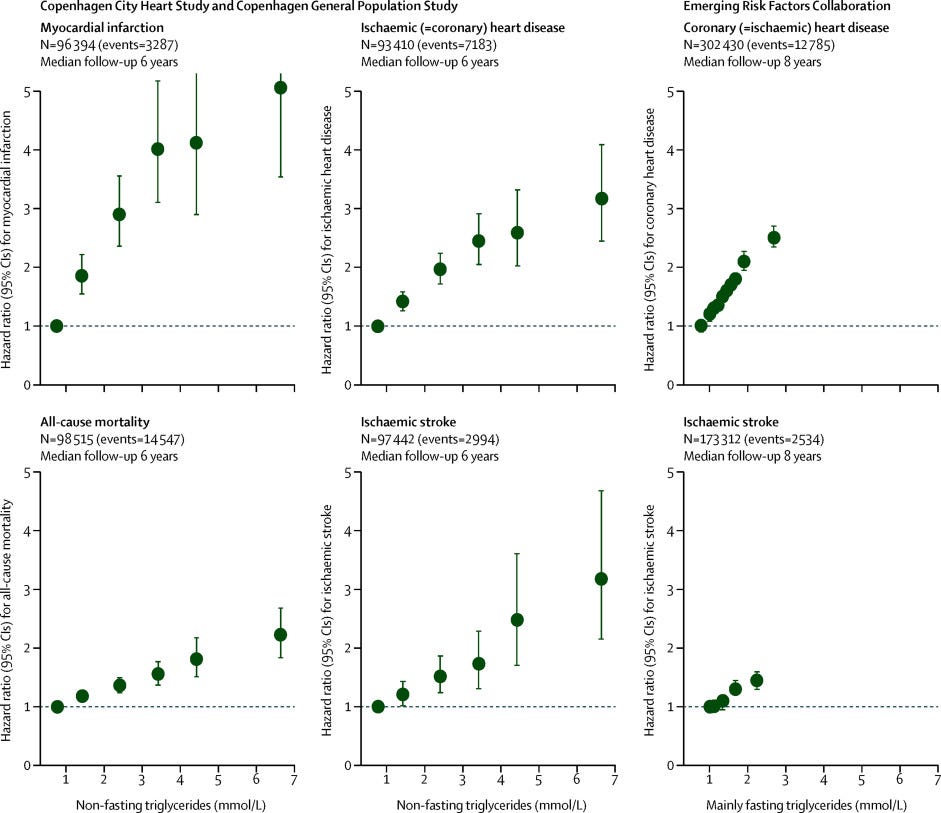
Despite these observations and high multipliers of risk, the epidemiological evidence wasn’t uniformly in favor of triglycerides as independent risk factors:
In 2009, the Emerging Risk Factors Collaboration that included 302 430 individuals from 68 long term, prospective studies, and 12 785 coronary events, similarly recorded that raised fasting and non-fasting triglycerides were associated with an increased risk of coronary (ischaemic) heart disease. This association was attenuated after adjustment for HDL cholesterol, and abrogated after additional adjustment for non-HDL cholesterok (cholesterol in LDL and remnants combined), in accordance with the idea that the cause of ischaemic heart disease is the cholesterol content in remnant particles, rather than raised triglycerides.
Nordestgaard et al. also comment on mendelian randomization studies and genome-wide association studies to conclude that:
…mendelian randomisation study with genetic variants in several candidate genes that affect the concentrations of remnant cholesterol or HDL cholesterol, or both, showed that an increase of 1 mmol/L in remnant cholesterol was associated with a 2.8-times increased risk of ischaemic heart disease that was not attributable to low HDL cholesterol concentrations; the corresponding observational risk was increased 1.4-times. Additionally, a doubling of genetically raised remnant cholesterol concentrations due to APOA5 genetic variants was associated with a 2.2-times increased risk of myocardial infarction, with a corresponding observational estimate of 1.7-times; for a genetically associated doubling in non-fasting triglycerides, the corresponding risk increases were 1.9-times causally and 1.6-times observationally.
Further,
…with use of genetic variants in LPL, a 1 mmol/L increase in triglycerides was associated with a 2.0-times increased risk of all-cause mortality, with a corresponding observational estimate of 1.2-times; or conversely, that a 1 mmol/L reduction in triglyceride concentrations was associated with a halved risk of all-cause mortality.
Finally,
…genetic studies strongly support the theory that high concentrations of triglyceride-rich lipoproteins or remnant cholesterol are causal risk factors for cardiovascular disease and all-cause mortality, and that low HDL cholesterol is probably an innocent bystander.
The risk posed by triglycerides is believed to be, based on our understatement of the pathophysiology of atherosclerosis and lipoprotein metabolism, mediated by an increase in remnant lipoprotein/cholesterol particles, making up VLDL and IDL particles.5 When triglycerides are high, the liver produces VLDL particles to traffic the triglycerides in. Eventually, lipoprotein lipase degrades the VLDL into IDL and hepatic lipase degrades the IDL further into LDL particles, depleting the particles of triglycerides at the same time.6 Increased triglycerides will increase the time VLDL and IDL particles, i.e. remnant lipoprotein particles, linger around in the bloodstream, thus contributing to atherosclerosis.
It’s important to realize that this surface-level explanation fails to capture the full complexity of the situation. Suffices to say that firstly, decreasing triglycerides will not necessarily decrease remnant lipoprotein particles, and may thus not contribute in any way to the absolute number of atherogenic particles. Secondly, the standard blood panel will fail to capture the complexity of the situation, even at the level described here. LDL-c only measures the cholesterol content of LDL particles, but will not inform the interpreter of the absolute number of LDL particles. Furthermore, triglycerides cannot be used as a proxy for the amount of VLDL +/- IDL particles, as it’s quite a stretch to believe that there would be any consistency between these on a population level. Thus we’d need a metric that can inform us about the absolute numbers of VLDL, IDL, and LDL particles, i.e. all the atherogenic particles. And as it happens, we do have one, namely, apoB. For a more comprehensive understanding of the issue, I’d recommend reading Hypertriglyceridemia and cardiovascular risk: a cautionary note about metabolic confounding.
To bring us back on track, contrary to the conclusion made by Nordestgaard et al., emerging evidence from more recent genetic studies, as well as previously mentioned PROMINENT, seems to dispute the causality of triglycerides in the development of CVD. In a mendelian randomization analysis7 from 2019 involving 654 783 participants, all the benefits of lowering triglycerides came via lowering of apoB:
The results of this study suggest that the clinical benefit of lowering triglyceride levels is similar to the clinical benefit of lowering LDL-C levels per unit change in ApoB and is proportional to the net absolute reduction in ApoB-containing lipoproteins. The results of this study therefore suggest that all ApoB-containing lipoprotein particles, including triglyceride-rich very-low-density lipoprotein (VLDL) particles and their metabolic remnants as well as LDL particles, have approximately the same effect on the risk of cardiovascular disease per particle. As a result, the clinical benefit of lowering triglyceride levels, lowering LDL-C levels, or lowering both may be proportional to the absolute change in ApoB-containing lipoproteins, regardless of the observed changes in plasma triglycerides or LDL-C.
The results of this study are consistent with the current understanding of the biology of lipids and atherosclerosis.
Further, data from 2 large observational cohort studies: the Atherosclerosis Risk in Communities (ARIC) study and the Framingham Offspring Study, seem to indicate that when “adjusting for non-HDL, the association between triglycerides and CVD events was no longer statistically significant (HR: 1.14 per doubling of triglycerides, 95% CI: 0.97–1.34).”8
As I’ve written earlier, apoB is the low-hanging fruit when assessing cardiovascular risk.9 There’s clearly an association between higher plasma triglycerides and increased apoB/non-HDL-c, making it important to distinguish between the causal factors, which should be the primary target of interventions, and the less causal factors that nevertheless might be indicative of the underlying pathophysiological processes and contribute to the primary causal factor.
Salim S. Virani discusses in his comments on PROMINENT how data from PROMINENT itself, as well as REDUCE-IT and STRENGHT, two major trials investigating the effects of EPA and EPA + DHA on cardiovascular events, seem to point in the direction of apoB being the desired target of intervention:
[in PROMINENT] Despite decreases in levels of remnant cholesterol, non-HDL cholesterol levels did not change significantly, whereas, as compared with placebo, the percentage change in the apolipoprotein B level was 4.8%. Although some of these findings would be expected given an increase in the low-density lipoprotein (LDL) cholesterol level with pemafibrate, they support the observation that in order for lipid-lowering therapies to show an effect, a net reduction in apolipoprotein B–containing lipoprotein levels is vital.

In the Reduction of Cardiovascular Events with Icosapent Ethyl–Intervention Trial (REDUCE-IT), apolipoprotein B levels were 9.7 percentage points lower and the incidence of primary end-point events was significantly lower in the icosapent ethyl group than in the placebo group. In contrast, the Outcomes Study to Assess Statin Residual Risk Reduction with Epanova in High Cardiovascular Risk Patients with Hypertriglyceridemia (STRENGTH) did not show a significant decrease in apolipoprotein B levels or a decrease in the incidence of cardiovascular events among patients who received Epanova, an eicosapentaenoic acid–docosahexaenoic acid combination. Furthermore, in REDUCE-IT, the decrease in the risk of atherosclerotic cardiovascular disease was not explained by triglyceride-lowering effects of icosapent ethyl. These results highlight the importance of net lowering of atherogenic lipoprotein levels rather than lowering of triglyceride levels per se.
The distinction between primary causal factors and secondary consequences of underlying processes is crucial when assessing the efficacy of an intervention. In high-risk patients, as stated earlier, apoB seems to be the desired primary target of intervention, while triglycerides in ranges below a significant risk of pancreatitis should be of secondary consideration. As concluded in the earlier comments on PROMINENT:
…fibrates should not be used to reduce the risk of atherosclerotic cardiovascular disease among statin-treated patients, although they may still have a role to play in decreasing the risk of pancreatitis associated with severe hypertriglyceridemia and perhaps nonalcoholic fatty liver disease. Alternatively, triglyceride lowering without decreases in the apolipoprotein B level will probably not suffice if therapies in development are to produce meaningful decreases in the risk of atherosclerotic cardiovascular disease.


